New Protocol
Data suggest that cell type, cell culture conditions, passage number, concentrations of stimuli, timing, and other factors have major implications for studies of Interleukin-11 (IL11) signaling in vitro. Primary cells, in particular, such as hepatic stellate cells, fibroblasts, and hepatocytes are ideal experimental systems. In contrast to immortalized cell lines, these primary cells better display relevant cellular physiology and pathobiology.
Although 2D cell cultures are the most common form of in vitro experimental systems, inasmuch as 2D is typically the easiest to handle and scale, growing cells on hard surfaces can induce mechanical stress, which has adverse effects on primary cells. 3D culture systems, and co-culture systems with several cell types, are viable alternatives. However, each approach comes with its own advantages and disadvantages. In a new series of protocols, the authors describe how to perform experiments with primary cells in 2D culture with minimal impact on cell physiology. The new protocols (see Viswanathan, S., Ng, B., Widjaja, A. A., Pua, C. J., Tham, N., Tan, J., Cook, S. A., & Schafer, S. (2021). Critical conditions for studying interleukin-11 signaling in vitro and avoiding experimental artefacts. Current Protocols, 1, e251. doi: 10.1002/cpz1.251) detail experimental and culturing conditions for primary cells that preserve meaningful cellular states and physiological responses ex vivo in conventional 2D cell culture systems.
Human Lung Fibroblasts
Pulmonary fibroblasts are a frequently used in vitro model system to study lung tissue repair and remodeling. HLFs can be isolated from healthy human lung tissue. These cells are adherent and adopt a spindle-like morphology when grown on plastic tissue culture dishes. Per the protocol, lung fibroblasts must be grown in monolayers before being stimulated with profibrotic cytokines. After passage two, cells can also be frozen and stored for future experiments. Cells should not be plated at lower densities than specified, which limit total cell numbers available for downstream experiments. The authors recommend measuring IL11RA protein expression to monitor primary cell integrity.
Primary Human Hepatic Stellate Cells (HSCS) and Human Renal Proximal Tubular Epithelial Cells (HRPTEpiCs)
HSCs are the precursor cells of fibrogenic myofibroblasts in the liver. Isolated cells are grown in a monolayer and should only be subjected to limited cell expansion. After passage two, cells can be frozen and stored for future experiments. Controlled population doublings are achieved by plating HSCs at the cell densities specified in the protocol, which also ensures that the cells are maintained at an exponential growth phase throughout the primary culture. Depending on the donor source and handling by commercial vendors, the passage number where cells are still suitable for IL-11 experiments can vary. To monitor primary cell integrity, the recommendation is to measure IL11RA protein expression.
Epithelial cells form a thin protective layer around the outer layers of body organs and vessels and express IL11RA. Similar to fibroblasts, they are frequently used in an in vitro model system to study tissue repair and remodeling. In disease, epithelial cells can be converted into stromal-like cells through a process called epithelial-mesenchymal transition (EMT). This disrupts secretion, sensing, selective absorption of the epithelial layer, and eventually impairs organ function. The cells in the protocol example are isolated from the human kidney, cryopreserved at passage one and supplied frozen. This protocol is optimized to reduce the loss of IL-11RA expression in vitro and again to monitor cell integrity the recommendation is to measure IL-11RA protein expression.
[button link=”https://currentprotocols.onlinelibrary.wiley.com/doi/10.1002/cpz1.251″ class=”short_code” newwindow=”yes”] Access the Publication[/button]
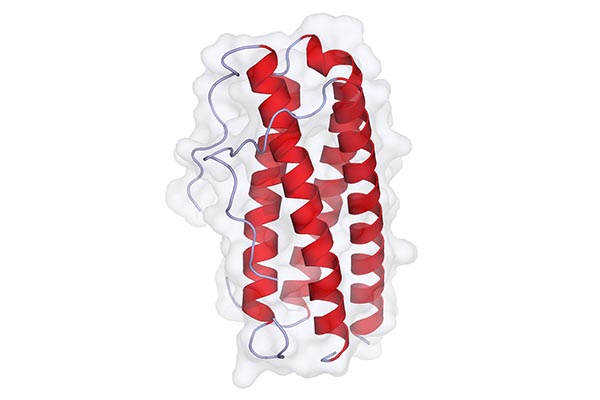
Feature Image By Agifr – Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=35656141