- Applications
Viable cell enrichment
Three-step cell enrichment in 20 minutes
The value of quality samples
In cellular biology, downstream assay results are highly dependent on the quality of the preliminary samples used. Poor quality samples – such as those containing a high percentage of dead cells and debris – will produce poor results. It is the fundamental quality of the starting sample that ultimately dictates the quality of the results, and it is the sample preparation step which is often the most difficult to achieve successfully.
By utilizing the LeviCell® platform for cell isolation, scientists have consistently demonstrated robust enrichment for a wide range of target cells – without affecting cellular expression or original population representation. This innovative Levitation Technology™ can be applied seamlessly to a wide range of research areas and workflows.
Preparation is key
Traditional methods used to separate specific cell types from a mixed population include magnetic beads, gradient purification, and flow cytometry. Although these techniques are widely used, they are not necessarily the most efficient or effective way to enrich for a target cell population.
Common issues that haunt scientists who use these methods include introducing artificial stress on cells that inevitably lead to an alteration in their expression profile (Van Den Brink et al., 2017), antibody-induced intracellular signaling (Kornbluth and Hoover, 1989; Christaki et al., 2011), as well as osmotic- and pressure-induced changes to the cell expression profile of multiple cell types (Xiong et al., 2002; Romero-santacreu et al., 2009; Van Den Brink et al., 2017).
These issues are not taken lightly, as they come at a price: the expenses associated with garnering difficult-to-obtain samples, materials, and labor; the immeasurable cost of frustration; and the irreplaceable expense of time. To this end, the LeviCell is a game changer.
Levitation Technology
LevitasBio® has developed an innovative levitation technology platform that uses less than 1 psi of pressure to enable a touch-free, label-free, three-step enrichment process that takes only 20 minutes to complete.
Live cell populations levitate much higher than dead cells and debris, allowing for clear separation and collection by the LeviCell, as shown. The fluorescent dyes used in the provided image were used solely to highlight the separation process and are not required to use the LeviCell system.
LeviCell’s gentle separation maintains maximum cell viability, enabling the preservation of cell population representation integrity for downstream assays such as single-cell analysis. The simple three-step process streamlines the workflow and minimizes the risk of contamination and damage from multiple instances of handling and washing, consequently maximizing enrichment for each cell type.
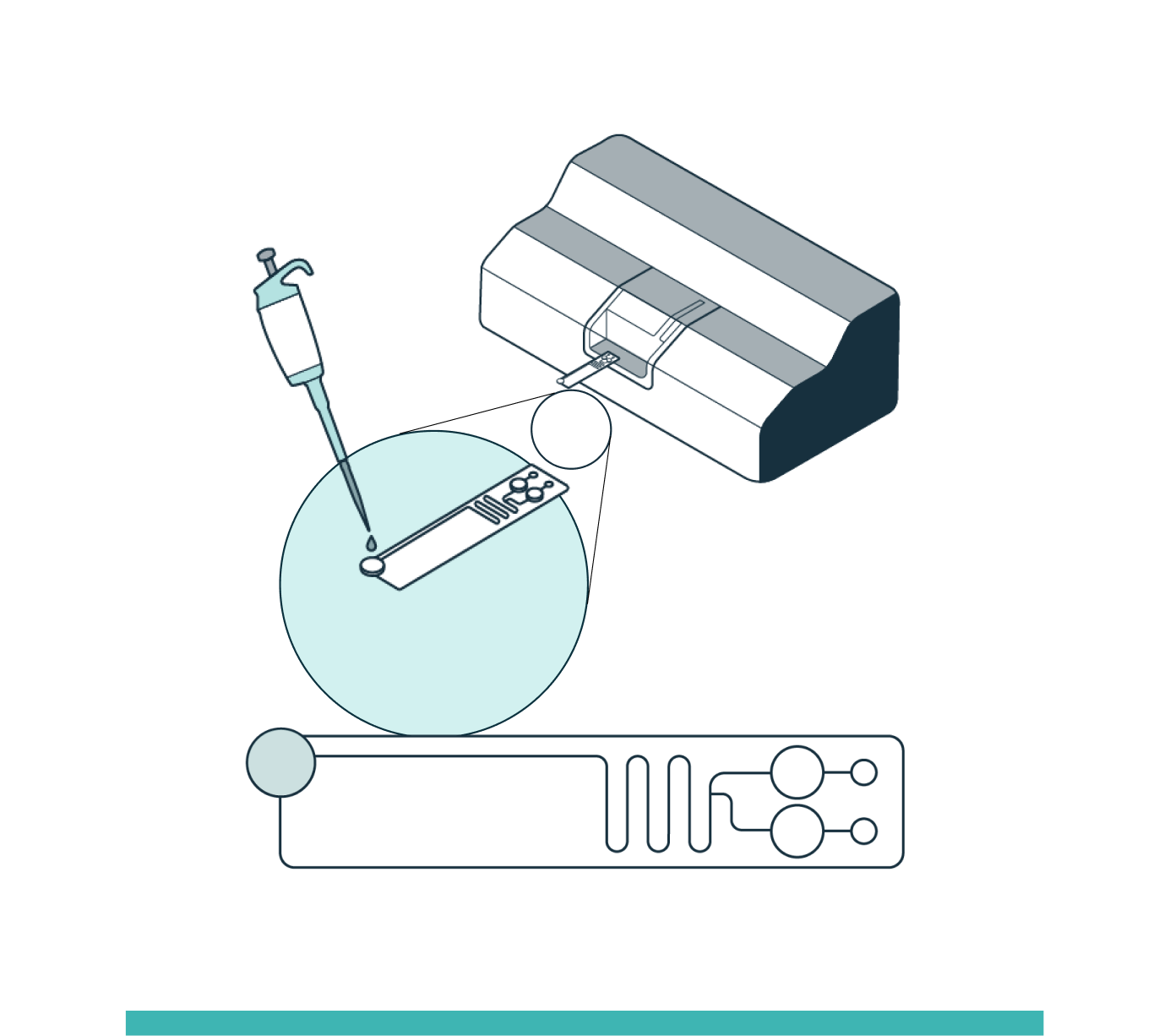
STEP 1
INPUT
Prepare the sample by adding the Levitation Agent, a paramagnetic solution. After inserting the cartridge into the system, dispense the sample into the inlet well.

STEP 2
ENRICHMENT
The system automatically loads the sample into the separation channel while the sample is imaged and levitated. The system performs automated, label-free enrichment plus debris removal via levitation.
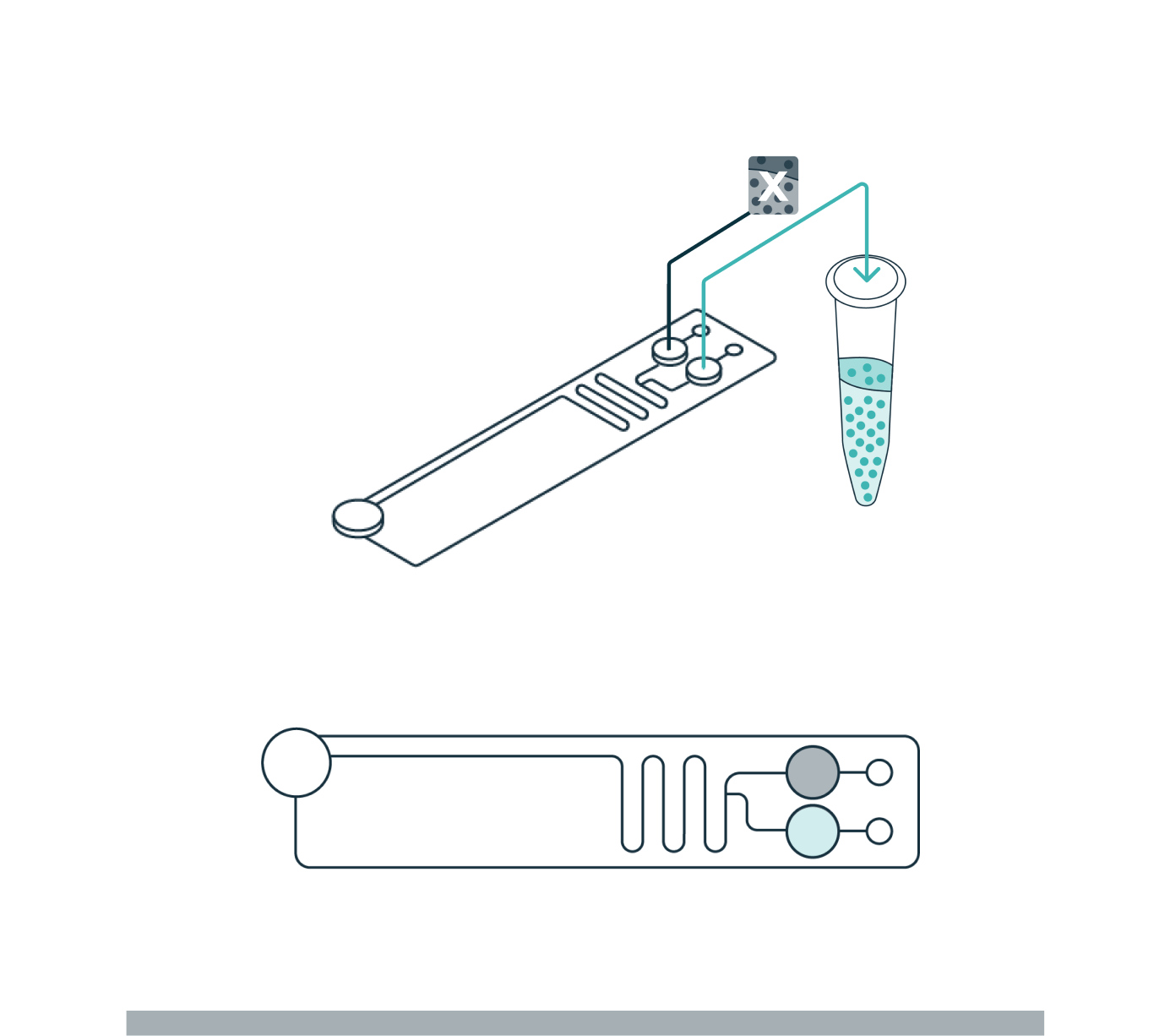
STEP 3
COLLECTION
Once the cells have levitated to their final equilibrium position, a live view in the software enables the user to define how the sample will be divided (fraction of interest and waste). Separated fractions are collected into the cartridge outlet wells.
Results
Primary samples were processed on the LeviCell system and using the most common bead-based approach from a leading supplier. The results presented here for both methods were achieved by manually counting the live and dead cells to calculate the yields and viabilities.
Revolutionary viability and yields
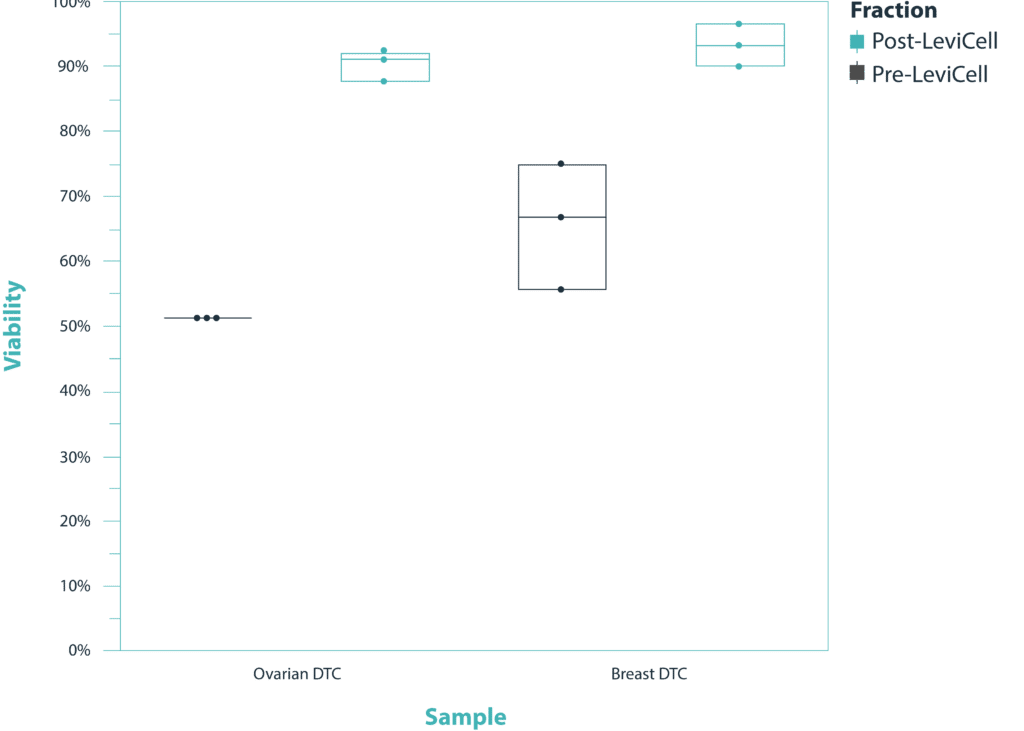
LeviCell demonstrated the successful removal of dead cells and debris from a variety of primary samples while maintaining the original representation of the populations found within the sample. Furthermore, the LeviCell was able to enrich a low starting number of live cells to a final viability of >90%, exhibiting a capacity for scalability regardless of starting number of cells. 100,000 cells from each of two dissociated tumor cells (DTCs) were successfully processed and enriched to a final viability of >90% through removal of dead cells and debris in 3 steps and 20 minutes.
Viability vs. Cancer & Fraction
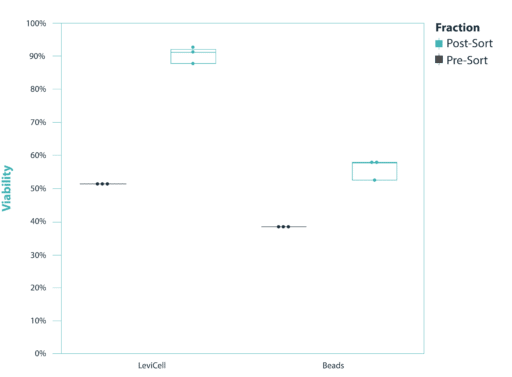
Superior performance over bead-based methods
The ovarian sample was split into two fractions of 100,000 cells and each fraction independently processed using either the LeviCell system or a popular bead-based dead cell removal kit. The output from each enrichment method was analyzed by manual cell counting using a Trypan Blue stain and a hemocytometer. LeviCell clearly outperforms the standard bead method in both Yield and Viability, while providing a faster, simpler and much more accommodating workflow when working with challenging samples. Additionally, LeviCell maintains the original cellular representation of the native sample without affecting the gene expression profile or activation state in even the most sensitive cell types.
Viability vs. Sort Method & Sort
Yield vs. Sort Method

Maximum levitation height difference between differentiated and undifferentiated adipocytes was achieved using 30 mM of Levitation Agent (Figure A). As the concentration of Levitation Agent increased, overall levitation height of both populations increased while the distance between differentiated and undifferentiated cells decreased.
No negative effects on post-processing population representation
The data on the right show how LeviCell’s gentle cell separation approach enables the study of notoriously delicate and sensitive primary cell types without compromising the integrity of the original cell populations. Sorted and unsorted ovarian DTC samples were blocked with TruStain human Fc Block for 15 minutes at RT then stained with 5 μL each of anti-CD45, anti-CD19, anti-CD11b, and 10 μL of anti-CD3, for 30 minutes at room temperature. Samples were washed with FACS buffer (0.5% BSA in PBS) once before analyzing on a Sony SH800S cytometer. The LeviCell output shows a similar amount of CD3+, CD19+, and CD11b+ cells within the CD45+ lymphocyte population while the sample processed by the bead method shows reduced proportions of CD3+ and CD11b+ cells.

No effect on the post-processing activation state
Macrophages, one of the most sensitive cell types, demonstrates the gentleness of the LeviCell system. The data below confirms there are no adverse side effects on cells’ post-processing activation state.
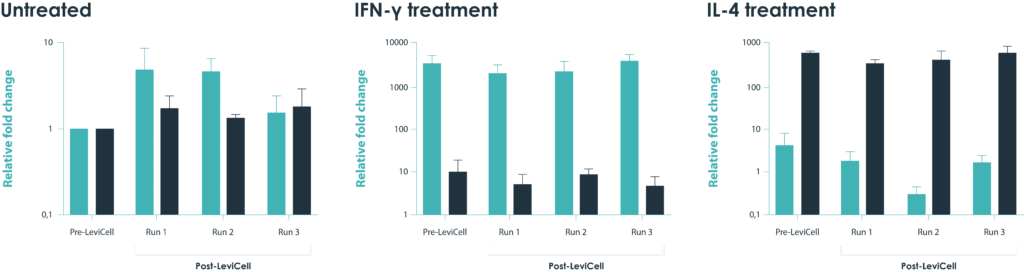
RAW264.7 cells were treated with IFN-g and IL-4 in order to polarize the macrophages into either the M1 or M2 stage. The samples were then processed on the LeviCell system. qRT-PCR was than performed to examine the expression of M1 specific gene (iNOS) or M2 specific (Arg1) in these cells to show that their activation states were not altered by the LeviCell. No differences in gene expression were observed between the input and output cells for the activation states.
LeviCell’s unique levitation platform enables scientists to significantly reduce costs (financial and otherwise) by reducing the amount of time to enrich for specific cell types from hours and days to mere minutes.
The three-step process simplifies the workflow and minimizes the need for highly skilled technicians and complicated single-cell preparation protocols. Because the LeviCell system does not use any high pressure, labels, dyes, or antibodies to isolate cells, alteration of expression profile. LeviCell’s closed environment also eliminates the risk of contamination and cell death due to manual manipulation. Separating live cells from dead and debris is as easy as finding the line of delineation between them.
