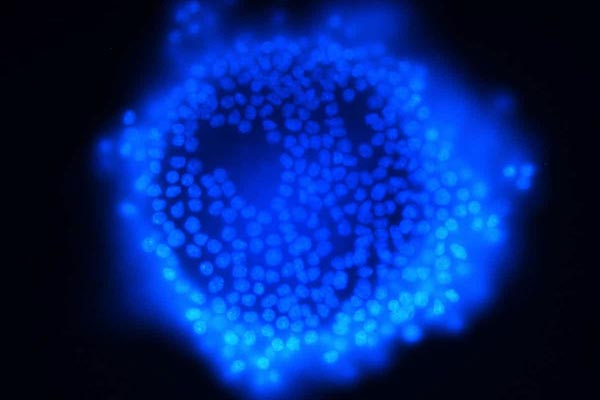
Oocytes are immature eggs that develop to maturity within the follicle. Granulosa cells (GC) provide the physical support and microenvironment required for the developing oocyte. These granulosa cells form a single layer around the oocytes in the primordial ovarian follicle and advance to form a multilayered cumulus oophorus surrounding the ovum in the follicle. The major functions of granulosa cells include the production of steroids and LH receptors.
GC apoptosis in ovulated cumulus‑oocyte complexes can be used as predictor of oocyte quality. Therefore, establishing appropriate in vitro GC models is important in the study of ovarian function and assists in elucidation of the mitogenic, luteotropic and apoptotic mechanisms responsible for selection and support of the dominant ovarian follicle.
Although several human GC isolation techniques have been described in the literature, only recently has a density gradient-based procedure been proven as a rapid, simple and relatively inexpensive technique. Writing in the September 2021 issue (Vol. 22, No. 3) of Experimental and Therapeutic Medicine, researchers Ying Han, Ge Gao, Shuang Li, Nan Xiao, Yinfeng Zhang, and Haining Luo (see Development of an optimal protocol for isolation and purification of human granulosa cells in patients with different ovarian reserves; DOI: 10.3892/etm.2021.10370) describe a new protocol to accurately and reproducibly isolate granulosa cells (GCs) in patients with different ovarian reserves. The cells of healthy individuals and patients with polycystic ovary syndrome (PCOS) were isolated using a modified two-step Percoll density gradient centrifugation. The cells of patients with poor ovarian response (POR) were isolated using a one-step method suitable for samples containing few cells.
Cells extracted using these purification techniques were compared regarding cell yield, viability and purity using immunocytochemistry, flow cytometry, Cell Counting Kit-8, western blotting and RNA integrity analysis. The purity and activity of the cells in the POR group were comparable with those in the healthy and PCOS groups and no statistically significant differences were identified. Furthermore, isolated cells analyzed for RNA integrity indicated good quality RNA and presented an RNA integrity number of 8-10, demonstrating that the technique enabled the isolation of GCs from different types of patients. Thus, this protocol demonstrates density-gradient based purification is a reliable and reproducible technique that enables the preparation of pure GCs with high yields for downstream evaluation of their molecular functions